Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoNew answer posted
7 months agoContributor-Level 10
Hess's law
ΔH_solution = ΔH_lattice + ΔH_hydration
4 = 788 + ΔH_hydration
ΔH_hydration = -784 kJ mol? ¹
New answer posted
7 months agoContributor-Level 10
In isoelectronic species nuclear charge can be approximated as
Nuclear charge ≈ z / no. of electrons
Al³? Mg²? Na? F? O²? N³?
Nuclear Charge: 13/10 12/10 11/10 9/10 8/10 7/10
Minimum nuclear charge is in N³? and maximum is in Al³?
So order should be
Al³? < Mg? < Na? < F? < O? < N?
New answer posted
7 months agoContributor-Level 10
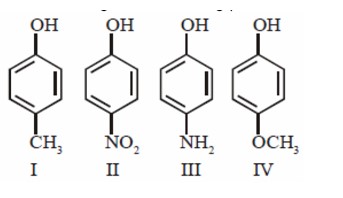
Using VSEPR, L.P - B.P repulsion we can safely say that CH? should have the highest bond angle among the given
New answer posted
7 months agoContributor-Level 10
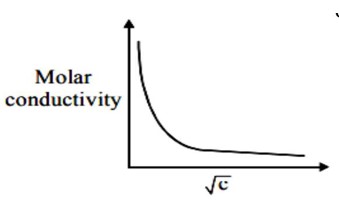
KNO? , HCl and NaCl are strong electrolytes for these electrolytes of Λ_m with √c will be liner which can be given as
Λ_m = Λ? _m - A√c for strong electrolyte
Since given variation is not linear it has to be a weak electrolyte
CH? COOH is a weak electrolyte
New answer posted
7 months agoContributor-Level 10
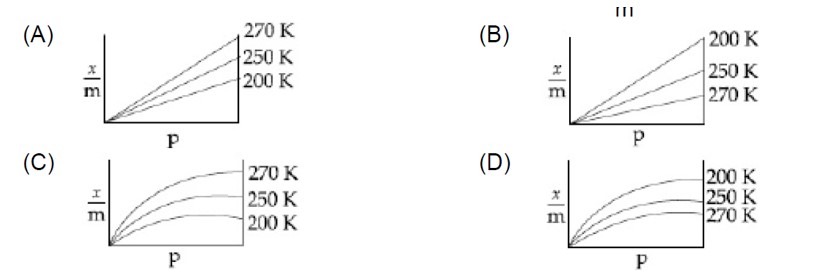
Gas + Solid? GS? H = -ve
Adsorbed gas
Adsorption of gas is an exothermic process. Increase in temperature reduces the extent of adsorption.
x/m = Kp¹/? (n > 1)
New answer posted
7 months agoContributor-Level 10
In FCC octahedral voids are present at the edge centers and body center
Consider a diagonal projected form edge centre passing through the body centre
Distance between octahedral voids = √2a/2 = a/√2
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers