Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
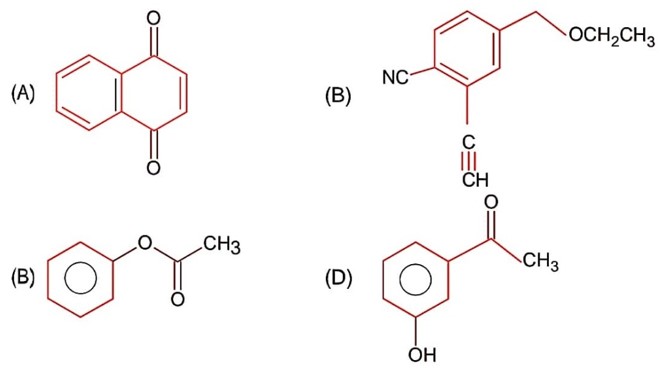
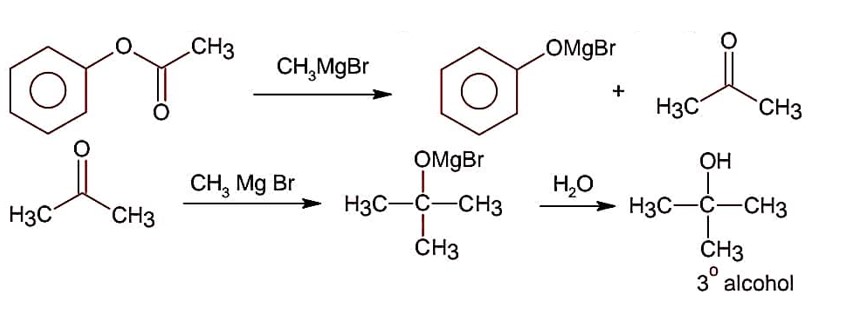
Esters on treating with excess CH3MgBr followed by hydrolysis gives 3° alcohol.
New answer posted
7 months agoContributor-Level 10
Electronic configuration of Ga+ ion = [Ar] 3d10 4s2 4p0
Last electron goes into S-orbital, hence
Azimuthal quantum number
New answer posted
7 months agoContributor-Level 10
NaOH + HCl -> NaCl + H2O
(milimole) t = 0 250 * 0.5 500 * 1.0 0 0
t =
New answer posted
7 months agoContributor-Level 10
vapour pressure of benzene at 20°C = 70 torr
= vapour pressure of toluene at 20°C = 20 torr
Mixture is equimolar, XB = 0.5 and XM = 0.5
Total vapour pressure (PT) = 70 * 0.5 + 20 * 0.5 = 45 torr
Mole fraction of benzene in vapour phase
= 0.777 = 77.7 * 10-2 torr
Ans. = 78 (the nearest integer)
New answer posted
7 months agoContributor-Level 10
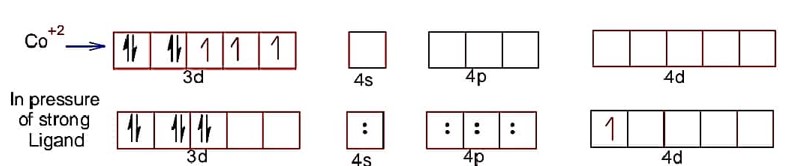
Hence, electronic configuration of CO2+ is
spin magnetic moment =
New answer posted
7 months agoContributor-Level 10
For Ni2+, crystal field CFSE magnitude shows the magnitude of absorbed light. Following are the energy absorbed order
Hence order of colour of compounds are.
New answer posted
7 months agoContributor-Level 10
Both statement are correct for the glass body heating, but reason is not correct explanation during heating process of glass, constituents unit rupture of glass body and gives the edge smoothness.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers