Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
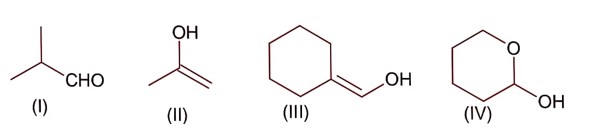
All structure can produce -CHO functional group which gives +ve test of Tollen's reagent except structure (II), because if forms ketonic group after tautomerisation
New answer posted
7 months agoContributor-Level 10
Deep blue colour solution
In cation analysis of Cu+ ions, precipitate formed is CuS on treating with H2S and HCl which dissolved in HNO3 and produced blue colour complex solution
New answer posted
7 months agoContributor-Level 10
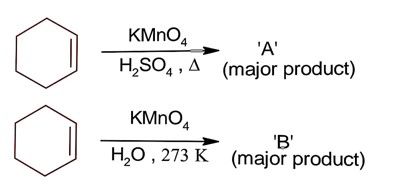
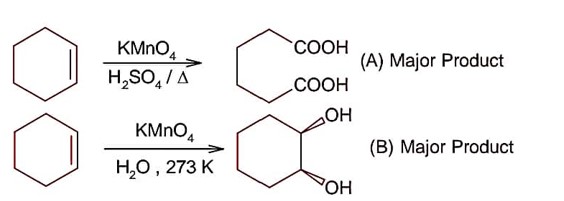
In
New answer posted
7 months agoContributor-Level 10
Mole of
1 mole of or KClO3 produced by 6 faraday (F).
mole of or KClO3 produced by (6 * 0.08156) F
New answer posted
7 months agoContributor-Level 10
= 0.0134 min-1
Rate constant at 300K = 1.34 * 10-2min-1 [the nearest integer = 1.0]
New question posted
7 months agoNew answer posted
7 months agoNew answer posted
7 months agoContributor-Level 10
In fcc structure of diamond four C present in fcc lattice and other four C present in tetrahedral voids where 50% of tetrahedral voids are occupied. Hence number of carbon atoms present per unit cell of diamond is 8.
New answer posted
7 months agoContributor-Level 10
Complex has Ni4+ and strong ligand, hence following are the metal ion electronic configuration
Change of unpaired electron = 2
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers